Abstract
Introduction: Acute myeloid leukemia (AML) patients with mutations in the FMS-like tyrosine kinase 3 (FLT3) gene represent 20-30% of all AML cases. These patients can be treated with tyrosine kinase inhibitors (TKIs) targeting FLT3; however, up to 60% of patients develop resistance or experience adverse side effects, which makes identifying mechanisms of drug resistance a priority. Our previous work demonstrated a tumor suppressor role for G0/G1 switch gene 2 (G0S2) in chronic myeloid leukemia (CML), which is profoundly downregulated in TKI resistance (Gonzalez MA, et al. under review). However, the role of G0S2 in FLT3+ AML is unknown. G0S2 regulates multiple cellular functions, including lipolysis, de novo lipogenesis, and oxidative phosphorylation; therefore, we hypothesized that reduced G0S2 expression in FLT3+ AML results in altered oxidative energy metabolism, promoting therapy resistance.
Methods: To address this hypothesis, we first used shRNA lentiviral vectors to transduce the FLT3+ AML cell lines, MOLM-13 and MOLM-14, with a non-targeting control vector (shNT), a G0S2 knockdown vector (shG0S2), and an ectopic G0S2 expression vector (pLVX G0S2). In primary FLT3+ AML MNCs, we used an empty vector (EV) and pLVX G0S2. We assessed colony formation ability by plating cells in 0.9% Methocult plus cytokines for primary cells, and measured apoptosis using AnnexinV and 7-aminoactinomycin D (7AAD). Cell lines were treated with Doxycycline (100 ng/ml) ± FLT3 inhibitors, either Quizartinib (20nM) or Midostaurin (50nM). We performed immunoblotting using SDS-page, PVDF membranes, and a human antibody cocktail for the 5-electron transport chain (ETC) complexes. For the in vivo experiments, NOD-scid IL2Rgammanull (NSG) mice received intravenous injections of 3 million cells/mouse of the MOLM-14 cell lines with the shNT, shG0S2, and pLVX G0S2 vectors, and all mice were immediately placed in doxycycline hyclate chow to induce vector expression. To assess the role of G0S2 in FTL3+ AML cells, we used the correlation feature available at UALCAN to calculate the genes co-expressed with G0S2 in The Cancer Genome Atlas (TCGA) AML data. Finally, we measured oxygen consumption rates (OCR) using the Cell Mito Stress Test kit for the Agilent Seahorse XFp Bioanalyzer.
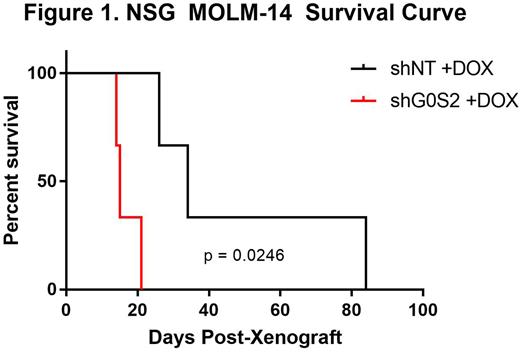
Results: Using publically available data, we found that G0S2 mRNA expression does not correlate with overall survival (OS) in AML, but is significantly downregulated in AML versus normal mononuclear cells (MNCs, p<0.001), and further reduced in AML patients with mutated FLT3 (p=0.006). In colony formation assays, the FLT3 TKI Midostaurin reduced colony number in the presence of G0S2 ectopic expression when compared to its control. In contrast, FLT3 TKIs had less of an effect on apoptosis in the presence of G0S2 ectopic expression. In primary FLT3+ AML MNCs, ectopic G0S2 expression resulted in a significant reduction of colony formation, which correlated with increased induction of apoptosis when compared to the empty vector control. Importantly, pathway enrichment analysis using data from The Cancer Genome Atlas (TCGA) revealed that the genes co-expressed with G0S2 in AML implicated a role in energy metabolism. After confirming G0S2 ectopic expression in the MOLM-13 cell line, immunoblotting showed reduced expression of complexes III and V of the ETC, which correlated with reductions in oxygen consumption rates using the Agilent Seahorse Bioanalyzer. In vivo, G0S2 knockdown in FLT3+ AML cell lines resulted in a significant reduction in overall survival of NSG mice compared with controls (Figure 1).
Conclusion: Altogether, our data demonstrate that loss of G0S2 expression in FLT3+ AML dysregulates oxygen metabolism by altering expression of ETC components. Ultimately, we showed that G0S2 is a tumor suppressor that is downregulated in FLT3+ AML, which might promote therapy resistance by altering oxidative energy metabolism.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal